This post is also available in:
 Italiano (Italian)
Italiano (Italian)
Presentation of Lavoisier’s Memoir : “EXPERIMENTS ON ANIMAL RESPIRATION” – year 1777
The main way to introduce the history and epistemology of chemistry into teaching is to use the memoirs, essays and pages of texts written by the protagonists of chemistry as a fundamental tool. I translated into english from French, I think for the first time, the memoir that Lavoisier read at the Academy of Sciences in 1777 entitled « Experiments on the respiration of animals and on the changes that take place in the atmospheric air that passes through their lungs» , because it constitutes his first organic attempt to question and overcome the theory of phlogiston.
The incipit of his memory:
« Of all the phenomena of animal economy, there is none more surprising, nor more worthy of the attention of physicists and physiologists, than those which accompany respiration. While we know little about the object of this singular function, we do know that it is so essential to life that it cannot be suspended for some time without exposing the animal to the danger of imminent death..»
With a series of experiments on elastic fluids (oxygen, carbon dioxide, nitrogen), which he carries out with a rigorous scientific method and which he describes with an effective and clear language, he is the first to establish the identity of the various elastic fluids of atmospheric air, through a process analogous to the analysis and synthesis of a compound, i.e. dividing the constituents of atmospheric air, and then reuniting them:
« This is the most complete type of proof that one can arrive at in Chemistry, the separation of air and its recomposition»
Then he investigates for the first time the changes that occur in the atmospheric air during respiration. But he also tries to answer the question of what happens in the lungs to cause this change. His insight into the pulmonary physiology of respiration takes us by surprise due to his depth. He also offers us an explanation of the color of blood which, while not entirely correct, is of great insight. We were saying that in the Memoir he questions the theory of phlogiston, and he does so by criticizing the interpretations of the experiments that Priestley, proponent of the phlogiston theory, had also conducted on elastic fluids.
“Furthermore, since I am about to combat Stahl’s doctrine of phlogiston with a series of experiments; the objections I will have against this doctrine will also fall on the flogistonized air, claimed by Mr. Priestley.”
The phlogiston theory will still have its influence. Lavoisier’s new ideas shake Stahl’s theory, but the prestige of the latter is still great. For many chemists, the path followed is not a radical choice of field, but a position of prudent waiting. This is confirmed by the author of the commentary on this Memoirs of Chemistry, whose name is not known, but he is certainly an authoritative member of the Academy:
« One of the more general chemical theories, received with minimal contrast, is that of phlogiston given by Stahl; but in almost all the phenomena where Stahl has seen a combination of phlogiston, there is the separation of a gaseous fluid: almost everywhere, according to Stahl, a body loses the phlogiston, a gaseous fluid combines with it; thus in the phenomena in which Staal has observed combinations or separations of phlogiston, we must also admit separations and combinations of air taken in the opposite direction. Stahl’s theory was therefore not complete, but had to be abandoned or only completed and corrected, and how was it to be corrected? These questions divide Chemists: only experience and time will have to decide them. Fortunately, it is less with reasoning than with experiments that physicists fight today; and these intense discussions, even if they had remained undecided, would at least have produced new facts, and would not have been useless for the sciences. Thus, even if the chemical doctrine that M. Lavoisier substitutes for that of Staal were rejected, these Memoirs would still be a collection of interesting facts that would serve for the progress of Chemistry.»
As J.H. Scott in his article on the J.C.E. “Qualitative adequacy of Phogiston”:
«The common treatment of phlogiston in basic chemistry textbooks gives the impression that it was a preposterous notion and that people were rather foolish to hold it»
in reality it was precisely its complexity combined with a certain versatility that made it an almost impregnable fortress for a long time.
The reproduction of the experiences of Memory in teaching
In a basic chemistry course, the contents of the Memoir do not present difficulties for the student, also considering that the experiments described are conducted with relatively simple equipment. In a basic chemistry laboratory some of his experiences can even be reproduced. Below is the apparatus for calcining mercury used in the experiences described in his Memoir .
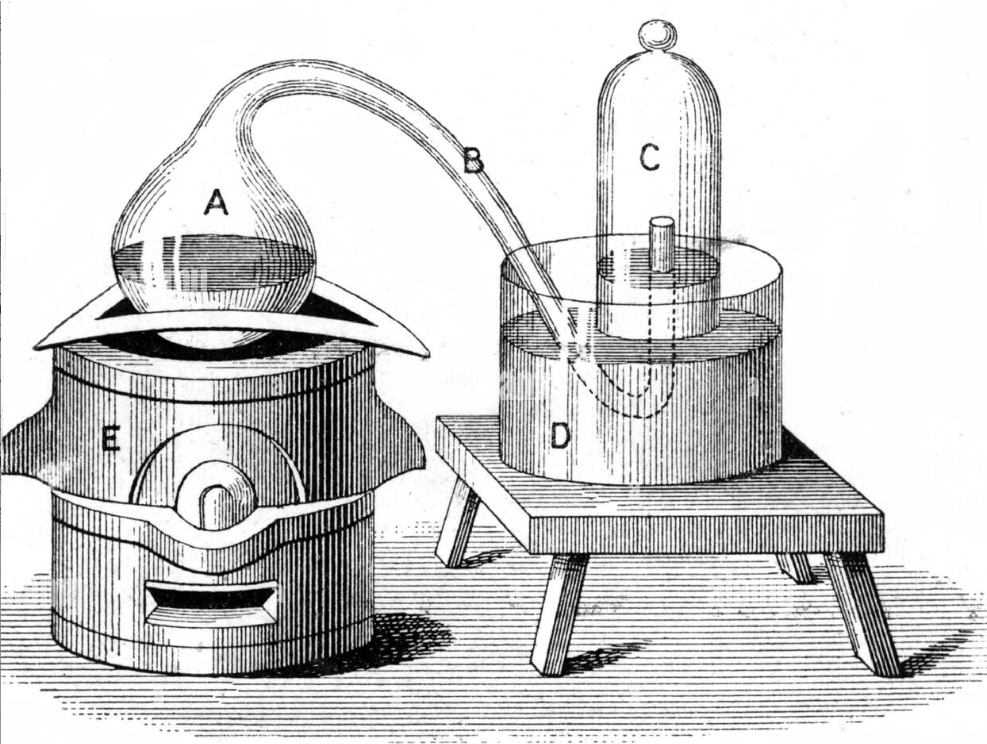
THE MEMORY OF LAVOISIER year 1777
This post is also available in:
 Italiano (Italian)
Italiano (Italian)

